 Note that in addition to the above two interactions, there is also a very strong inverted nuclear spin-orbit interaction, which splits the spherical symmetry and produces energy levels with strong $j$-dependence, where $j$ is the total angular momentum of a nucleon. The nuclear radius $r_0 A^$ is very nearly the same for both species. Note that the nuclear potential is spherical, so the split in the middle is meant to cleanly separate the radial shape of the proton and neutron potentials. This is well illustrated in the figure below, which sketches the filling of (not realistic) nuclear levels for a nucleus with 6 protons and 6 neutrons. Whereas neutrons are not subject to a Coulomb force, protons are so, as a result of the Coulomb repulsion, the energy levels of protons are typically higher than those of the neutrons, and the proton part of the potential also has a " tunnelling lip". This is the average potential felt by one nucleon and generated by all other nucleons, irrespective of species. As a result, both species live in a common potential knows as Wood-Saxon (or inverted Fermi) potential. The nuclear force is largely independent of the electric charge and affects neutrons and protons in approximately the same wave. the exclusion is on identical protons by themselves, and on identical neutrons by themselves. Neutrons are certainly distinguishable from protons, and both separately satisfy the Pauli exclusion principle, i.e. That means we have to accept that the shell model may be a good qualitative description, but we have to be cautious about pushing it further than that. This effect is small enough to be ignored (mostly) in atoms, but in nuclei the nucleons are so close that the nuclear orbitals are heavily mixed. However the electron-electron pair repulsion mixes up the atomic orbitals so strictly speaking they don't exist as individual separate orbitals. The atomic orbitals we all know and love, the $1s$, $2s$, etc, appear in an approximation known as the mean field. I say to a reasonable approximation because neither nuclear orbitals nor atomic orbitals really exist. Note that the protons and neutrons have their own separate sets of orbitals. The exclusion principle applies to all fermions, including protons and neutrons, so the protons and neutrons pair up two per orbital, just as electrons do. This description of the nucleus is known as the shell model. To a reasonable approximation the protons and neutrons in a nucleus occupy nuclear orbitals in the same way that electrons occupy atomic orbitals. What happens when we consider more than 2 protons/neutrons? The only explanations I've been able to find consider 2 protons and state that they can have different spin. 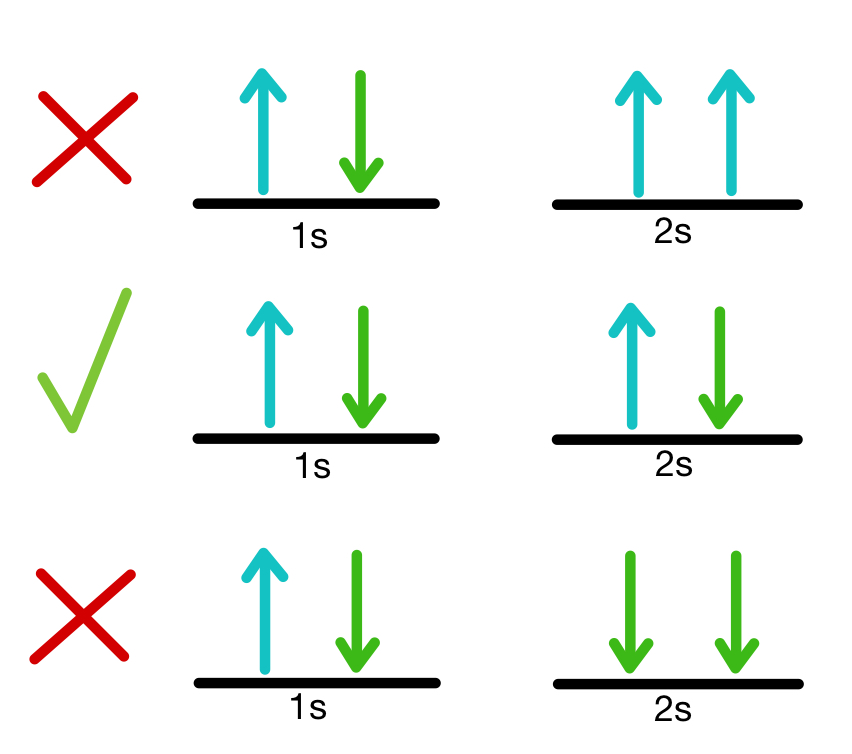
I super confused about how the exclusion principle works for protons and neutrons. In other words, (1) no more than two electrons can occupy the same orbital and (2) two electrons in the same orbital must have opposite spins ( Figure 46 (i) and (ii) ). I understand that neutrons and protons are not identical fermions but considering them individually, suppose in a nucleus with X protons, are the energies of individual protons different from one another (and similarly for neutrons in the nucleus)?Īpologies, I'm not very familiar with quantum theory or the maths involved. Paulis Exclusion Principle states that no two electrons in the same atom can have identical values for all four of their quantum numbers. Protons and neutrons are both fermions, so why in a nucleus can multiple protons and neutrons simultaneously exist. I understand this for electrons that for each energy level in an atom there are two possible electrons that may occupy this energy state but with opposite spin numbers. Wikipedia states it as "two identical fermions cannot occupy the same quantum state in a quantum system". I'm very confused about the Pauli exclusion principle. 
This means that one electron has an up spin of $ \dfrac$. The set of all these quantum numbers for electrons is different as electrons are present in an orbital with opposite spins. There are 4 quantum numbers, principle (n), azimuthal (l), magnetic (m), and spin (s). This principle can also be defined in another way as no two electrons in an atom can have the same set of all the four quantum numbers. This means that one orbital contains only two electrons that have opposite spin. It has made rules on accommodation of electrons in any orbital.Īccording to Pauli’s exclusion principle, any atomic orbital can not accommodate more than 2 electrons with the opposite spin. Pauli's exclusion principle tells us that these sets of quantum numbers should be different in the filling of electrons. Hint: The filling of electrons is done into the orbitals which are s, p, d, f. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
